 Long exposure can even cause coma or death. Due to this, nausea, vomiting, and bleeding may occur. It forms alloys with alkali metals, gold and mercury. It is outstanding in keeping time with precision, so it is used in atomic clocks. Radioactivity: If a person comes in contact with a radioactive isotope of caesium then cell-damage may occur. Periodic Table Definition, mass & chemical names Caesium Caesium, also spelt as Cesium, was discovered in 1860. Its advantages are that it has a half-life of 30 years and it has barium-137 as a stable end product. Nuclear application: Caesium-137 is used as a gamma emitter in industrial applications.Molecular Biology: High density caesium compound solutions such as caesium chloride, caesium sulfate, etc are used in density gradient ultracentrifugation.Atomic-clocks: They use the electromagnetic transition in the hyperfine structure of caesium-133 atoms as the reference point. 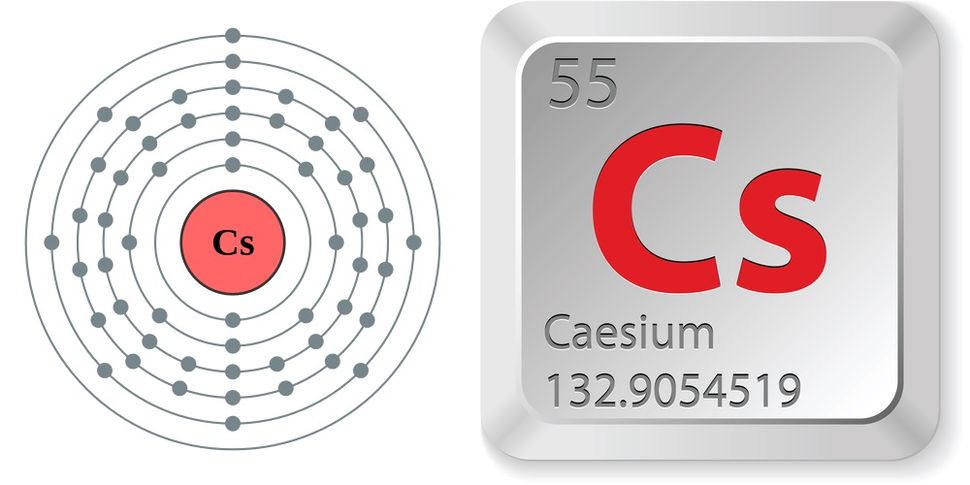
It also helps bring rock cuttings to the surface and to maintain pressure on the formation during drilling of the well. The main function of drilling fluids is to lubricate drill bits. The cesium-137 capsule, part of a density gauge used at Rio Tintos Gudai-Darri mine in Western Australia, was found after a survey vehicle travelling at 70. Petroleum exploration: The largest use of caesium is as a drilling fluid for extractive oil and petroleum industry. Relevance in Chemical and Related Industries This precipitated double salt is then decomposed and the water is evaporated to obtain pure caesium chloride. The grounded ore is dissolved in strong acids which leads to formation of caesium double salts. The caesium is extracted from pollucite mainly by acid digestion. The ore is crushed, hand-sorted, and then grounded. Caesium forms variety of compounds including variety of halides and oxides.įrom Pollucite: The mining and refining of pollucite is a selective process hence it is done on a smaller scale.Caesium can only be handled under inert atmosphere.It even reacts with ice whose temperature is as low as -116☌. The aim of present study was to compare the biological activity of plasmid purified by CsCl2 buoyant density gradient centrifugation and anion exchange HPLC. It reacts explosively with water even at low temperatures. Caesium is pyrophyric in nature, which means that it spontaneously ignites in air.Among them, the only stable isotope is caesium-139. Caesium is softest among all metals which are solid at room temperature.The density of caesium is 1900 in S.I.Caesium is a soft metal which shines in gold colour.It is a relatively rare element and makes up 3 ppm of Earth’s crust. Cesium chloride is used in centrifugation, a process that uses the centrifugal force to separate mixtures based on their molecular density. This citation format is based on MLA.Caesium is found in minerals pollucite and lepidolite. Also replace URL for the actual url of this page (The stay, ok?). 
Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. "Density of Cesium (Cs) [& g/cm3, kg/m3, Uses, Sources. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: That gives credibility to your paper and it is sometimes required in higher education. Want to learn more details and data about Cesium (Cs)? Check my Elements Comprehensive List.Ĭitation When you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. Name OriginLatin: coesius (sky blue) for the blue lines of its spectrum.ĭiscoveryDiscovered By: Gustov Kirchoff, Robert BunsenĭescriptionVery soft, light gray, ductile metal. SourcesFound in pollucite and as trace in lepidolite. Since it ionizes readily, it is used as an ion rocket motor propellant. 
Also used in producing photoelectric devices and atomic clocks. UsesUsed as a 'getter' to remove air traces in vacuum and cathode-ray tubes. In the case of Cesium, density is 1.873 g/cm³. However, there are interesting facts about Cesium that most don't know about. But what is the density of an atom of Cs? Radioactive check radioactive density meter caesium-137 and cobalt-60 radioactive density gauge for sale, You can get more details about from mobile site on. So it is expected that the smallest piece of matter, like an atom of Cesium has it too. Everything has mass and volume, and therefore density. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
